Liofilchem® Furazolidone FR, Antimicrobial Susceptibility Discs, 50 µg
Catalog No :
CAS Number :
Brand :
In Stock
Specifications:
| Application | Antimicrobial Susceptibility and Resistance Testing (AST) |
| Storage Temperature | -20°C |
| Product Type | Antibiotic Disc |
| Product Brand | Liofilchem |
| Product Grade | Microbiology grade |
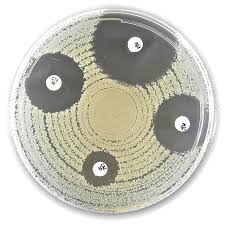
Liofilchem® Furazolidone FR Antimicrobial Susceptibility Discs are standardized antibiotic-impregnated paper discs used for the in vitro determination of bacterial susceptibility to Furazolidone by the disk diffusion method.
Furazolidone is a nitrofuran antimicrobial agent historically used against a range of Gram-negative and Gram-positive bacteria, particularly in enteric infections. These discs are intended for routine clinical microbiology testing, epidemiological surveillance, research, and teaching applications.
Each disc is manufactured under strict quality-controlled conditions, ensuring accurate antibiotic content, reproducible diffusion characteristics, and reliable zone diameter results.
Intended Use
- In vitro antimicrobial susceptibility testing (AST)
- Determination of bacterial susceptibility or resistance to Furazolidone
- Support of clinical diagnosis, alongside clinical and laboratory findings
- Research and teaching in microbiology laboratories
Principle of the Test
The disk diffusion method involves placing a Furazolidone-impregnated disc onto the surface of an agar plate (typically Mueller–Hinton Agar) previously inoculated with a standardized bacterial suspension.
- Furazolidone diffuses radially into the agar
- Susceptible organisms show inhibition of growth, forming a clear zone
- The diameter of the inhibition zone is measured and interpreted using accepted interpretive criteria, where available
Product Specifications
| Parameter | Specification |
|---|---|
| Antibiotic | Furazolidone (FR) |
| Disc Content | 50 µg |
| Test Method | Disk diffusion |
| Format | Paper discs |
| Intended Use | AST (in vitro diagnostic use) |
| Storage | As per label instructions |
| Manufacturer | Liofilchem®, Italy |
Available Formats & Ordering Information
| Reference | Format | Description | Packaging |
|---|---|---|---|
| 9099 | Cartridge | Furazolidone FR 50 µg | 5 × 50 discs |
| 9099/2 | Canister | Furazolidone FR 50 µg | 250 discs |
Quality Assurance
- Controlled antibiotic content per disc
- Batch-to-batch consistency verified
- Suitable for use with quality control strains
- Manufactured according to international microbiology standards
Applications
- Clinical microbiology laboratories
- Hospital diagnostic laboratories
- Veterinary and food microbiology (research use)
- Academic and teaching laboratories
- Antimicrobial resistance (AMR) studies
Storage & Handling
- Store under conditions specified on the product label
- Avoid moisture and excessive heat
- Do not use beyond expiry date
- Allow discs to equilibrate to room temperature before use
Limitations
- Results must be interpreted using recognized standards, where applicable
- AST results should be correlated with clinical context
- Furazolidone use may be restricted or regulated in some regions
Liofilchem® Furazolidone FR Antimicrobial Susceptibility Discs (50 µg) provide a reliable and standardized tool for evaluating bacterial susceptibility to Furazolidone using the disk diffusion method. Available in both cartridge and canister formats, these discs are ideal for routine testing, surveillance, and microbiology education, ensuring consistent and reproducible AST results.
- package Type: In Cartridge In Canister




 0
0