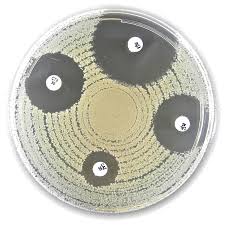
Liofilchem® Meropenem MRP, Antimicrobial Susceptibility Discs, 10 µg
Catalog No :
CAS Number :
Brand :
In Stock
Specifications:
| Application | Antimicrobial Susceptibility and Resistance Testing (AST) |
| Storage Temperature | -20°C |
| Product Type | Antibiotic Disc |
| Product Brand | Liofilchem |
| Product Grade | Microbiology grade |
Liofilchem® Meropenem MRP 10 µg Antimicrobial Susceptibility Discs are standardized in vitro diagnostic tools designed for determining bacterial susceptibility to meropenem, a broad-spectrum carbapenem antibiotic. These discs are used in the Kirby-Bauer disk diffusion method, one of the most widely accepted techniques in clinical microbiology laboratories for antimicrobial susceptibility testing (AST).
Meropenem discs play a critical role in detecting carbapenem-resistant bacteria (CRB) and identifying potential carbapenemase-producing organisms (CPOs)—key threats in hospital-acquired infections and multidrug-resistant (MDR) pathogens.
Key Features
- Standardized Potency: Each disc contains 10 µg of meropenem for consistent and reproducible testing
- High Manufacturing Quality: Produced under strict quality control to ensure batch uniformity
- Accurate Resistance Detection: Supports identification of carbapenem-resistant Gram-negative bacteria
- Guideline Compliant: Designed for use in accordance with EUCAST and CLSI standards
- Easy Workflow Integration: Compatible with standard Mueller-Hinton agar and disc dispensers
Clinical Applications
1️⃣ Routine Antimicrobial Susceptibility Testing (AST)
Used in hospital and reference microbiology laboratories to determine susceptibility profiles of Gram-negative organisms such as:
- Escherichia coli
- Klebsiella pneumoniae
- Pseudomonas aeruginosa
- Acinetobacter baumannii
Results guide clinicians in selecting effective antimicrobial therapy.
2️⃣ Detection of Carbapenem Resistance
A reduced or absent inhibition zone may indicate carbapenem resistance and possible carbapenemase production. These discs are often used alongside confirmatory methods such as:
- Carba NP test
- Modified Hodge Test (MHT)
- Molecular carbapenemase assays
3️⃣ Epidemiological Surveillance
- Supports antimicrobial stewardship programs (ASP)
- Assists infection control teams in tracking resistance trends
- Useful in research and public health laboratories
Application Procedure (Kirby-Bauer Method)
- Prepare a 0.5 McFarland standardized bacterial suspension
- Inoculate Mueller-Hinton agar uniformly
- Place the Meropenem 10 µg disc on the agar surface
- Incubate at 35 ± 2°C for 16–20 hours
- Measure the inhibition zone diameter
- Interpret results according to current EUCAST or CLSI breakpoints
Packaging Information
- 50 discs per cartridge
- 5 cartridges per pack
- Compatible with Liofilchem® disc dispensers
Storage & Stability
- Store at 2–8°C (Refrigerated)
- Keep sealed and protected from moisture and light
- Do not use expired or compromised discs
Regulatory Information
- For In Vitro Diagnostic (IVD) Use Only
- CE-marked
- Designed for use in compliance with EUCAST and CLSI guidelines
Liofilchem® Meropenem MRP 10 µg Discs provide reliable and standardized antimicrobial susceptibility testing for clinical microbiology laboratories. Their role in detecting carbapenem resistance makes them essential tools in infection control, antimicrobial stewardship, and resistance surveillance programs.
Ideal for hospitals, diagnostic laboratories, research institutions, and public health facilities requiring accurate carbapenem susceptibility testing.




 0
0