Q-Plex™ Human Cytokine Inflammation Panel 1 (9-Plex)
Catalog No :
CAS Number :
Brand :
In Stock
Target Analytes (9-Plex Panel)
Interferon Gamma (IFNγ)
Interleukin-1 Alpha (IL-1α)
Interleukin-1 Beta (IL-1β)
Interleukin-2 (IL-2)
Interleukin-4 (IL-4)
Interleukin-6 (IL-6)
Interleukin-8 (IL-8)
Interleukin-10 (IL-10)
Tumor Necrosis Factor Alpha (TNFα)
Specifications:
| Application | Protein Assays & Analysis | ||
| Storage Temperature | 2-8°C | ||
| Product Type | Assay Kits | Forms | Kit with Various components |
| Product Brand | QUANSYS Biosciences | ||
| Product Grade | Molecular Biology | ||
The Q-Plex Human Cytokine Inflammation Panel 1 (9-Plex) is a fully quantitative, planar-based multiplex ELISA designed for the simultaneous detection of nine key human inflammatory cytokines in serum or plasma samples. This chemiluminescent assay provides high sensitivity, broad dynamic range, and excellent reproducibility, enabling comprehensive cytokine profiling in a single well using only 25 µL of sample.
The assay allows up to 80 samples to be analyzed per 96-well plate within approximately 2.25 hours, delivering time-efficient and cost-effective multiplex performance compared to traditional single-analyte ELISAs.
Target Analytes (9-Plex Panel)
- Interferon Gamma (IFNγ)
- Interleukin-1 Alpha (IL-1α)
- Interleukin-1 Beta (IL-1β)
- Interleukin-2 (IL-2)
- Interleukin-4 (IL-4)
- Interleukin-6 (IL-6)
- Interleukin-8 (IL-8)
- Interleukin-10 (IL-10)
- Tumor Necrosis Factor Alpha (TNFα)
Assay Principle
The Q-Plex platform uses a planar multiplex format in which nine distinct capture antibodies are precisely adsorbed in a defined spatial array at the bottom of each well. During incubation, target cytokines bind to their respective capture antibodies. A detection antibody mix followed by Streptavidin-HRP enables signal generation.
Upon addition of chemiluminescent substrate, emitted light is captured and quantified using a compatible Q-View imager and analyzed with Q-View Software. The assay delivers highly quantitative results with less than 1% intra-panel cross-reactivity.
Performance Specifications
| Analyte | Calibrator Range (pg/mL) | ULOQ (pg/mL) | LLOQ (pg/mL) | LOD (pg/mL) | Inter-Assay CV | Intra-Assay CV | Avg. Linearity |
|---|---|---|---|---|---|---|---|
| IFNγ | 8000 – 10.97 | 5600 | 14.3 | 5.49 | 8% | 4% | 97% |
| IL-1α | 4000 – 5.49 | 2800 | 7.1 | 2.74 | 6% | 7% | 86% |
| IL-1β | 12226 – 16.77 | 8558 | 21.8 | 8.39 | 8% | 5% | 94% |
| IL-2 | 5213 – 7.15 | 3649 | 9.3 | 3.58 | 6% | 4% | 99% |
| IL-4 | 1676 – 2.30 | 1173 | 3.0 | 1.15 | 6% | 6% | 94% |
| IL-6 | 3000 – 4.12 | 2100 | 5.3 | 2.06 | 7% | 4% | 104% |
| IL-8 | 2000 – 2.74 | 1400 | 3.6 | 1.37 | 12% | 7% | 100% |
| IL-10 | 8039 – 11.03 | 5627 | 14.3 | 5.51 | 7% | 5% | 103% |
| TNFα | 6500 – 8.92 | 4522 | 11.5 | 4.46 | 6% | 5% | 98% |
Intra-Assay Cross-Reactivity: All analytes <1%
Validated Sample Types
- Human Serum
- EDTA Plasma
- Heparin Plasma
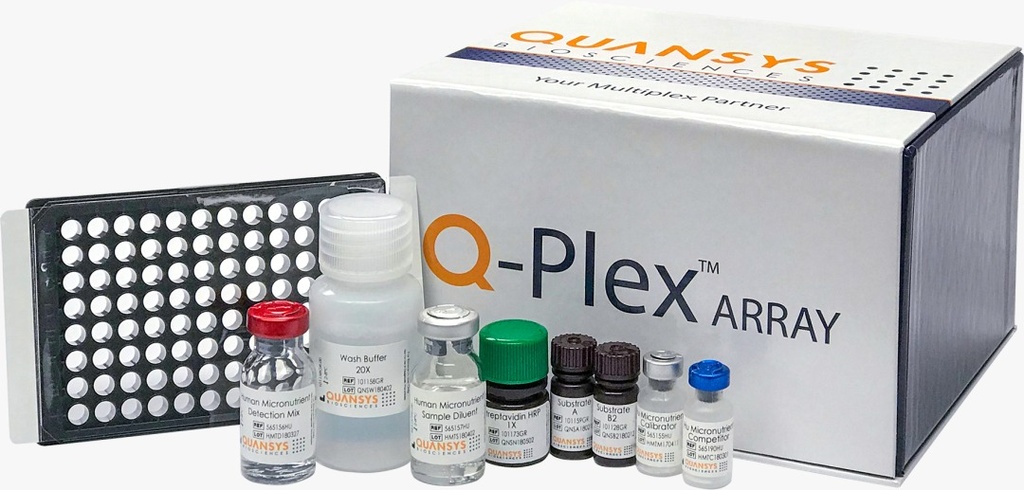
Kit Contents
Each kit includes:
- 96-well plate pre-coated with the 9-plex cytokine panel
- Calibrator standards
- Detection mix
- Streptavidin-HRP
- Chemiluminescent substrates
- Sample diluents
- Wash buffer
- Instruction manual
Required Instrumentation and Software
All Quansys Q-Plex multiplex assays require the use of Q-View imagers for signal detection and Q-View software for data acquisition and analysis. A fully functional trial version of the software is available, after which a licensed version is required. Performance with third-party imaging systems is not guaranteed.
Applications
- Cytokine profiling in inflammation studies
- Autoimmune disease research
- Infectious disease and sepsis monitoring
- Vaccine immunogenicity evaluation
- Cancer immunology and immunotherapy research
- Translational and biomarker discovery studies
The Q-Plex Human Cytokine Inflammation Panel 1 (9-Plex) provides high analytical sensitivity with low limits of detection across all analytes. The planar multiplex format eliminates bead handling and flow cytometry requirements while maintaining strong precision and minimal cross-reactivity. The small sample requirement makes it suitable for limited-volume clinical samples, and the rapid workflow supports high-throughput laboratory environments.




 0
0